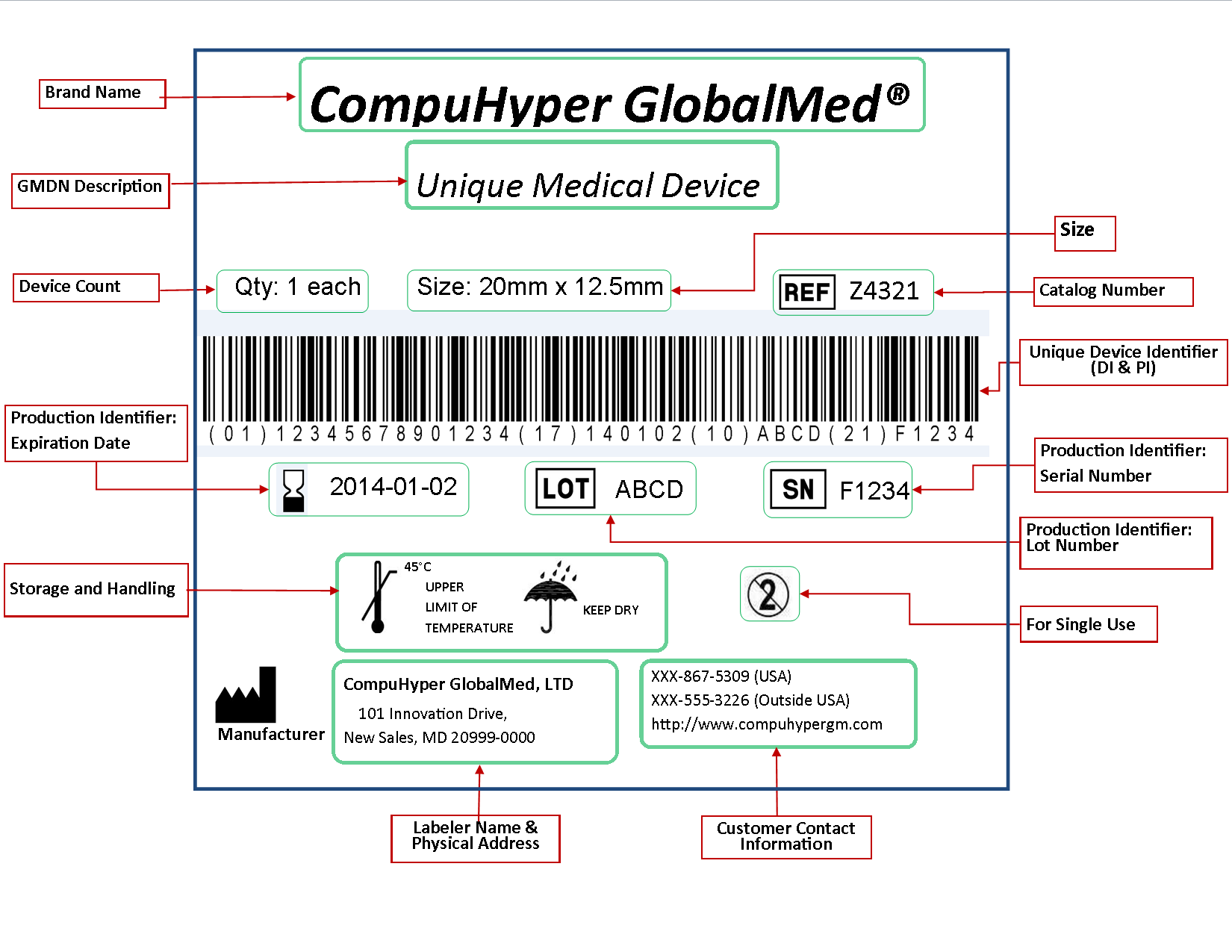
The Global Unique Device Identification Database (GUDID) contains key device identification information submitted to the FDA about medical devices that have Unique Device Identifiers (UDI).
The FDA is establishing the unique device identification system to adequately identify devices sold in the U.S.- from manufacturing through distribution to patient use. You can use AccessGUDID to search for specific medical devices or download all the GUDID data at once. We anticipate the release of additional web services for testing by the end of 2015. Please see the API Documentation for more information.
Medical devices cover a wide range of products - implants, CT scanners, surgical instruments, contact lenses, wheelchairs, and blood glucose tests, to name a few. Unlike drugs, many medical devices currently do not have a unique identifier that clearly distinguishes one product from another. But this is changing. The UDI system will provide a consistent and standard way to identify medical devices throughout their distribution and use by health care providers and patients.
Most devices will be required to have a UDI on their label and packaging, and for certain devices, on the product itself. Device companies must also submit certain information about these devices to the GUDID (pronounced "Good ID"). Please note: The GUDID does not collect or contain any information about patients or anyone who uses a device.
UDIs will be phased in over several years, starting with the highest risk devices, such as heart valves and pacemakers. Because of this, records for only a fraction of devices currently in use have been submitted to GUDID. For more information about UDI and GUDID, see below.
The National Library of Medicine (NLM), in collaboration with the FDA, has created the AccessGUDID portal to make device identification information in the GUDID available for everyone, including patients, caregivers, health care providers, hospitals, and industry. In our beta release of AccessGUDID, basic search and download functions are available. We plan to develop advanced search and web service capability in the future. The device information available on AccessGUDID is the most recent data submitted to the FDA that has completed the "grace period" after initial publication. (The grace period is the time during which device companies may make significant edits to their information; once the grace period is completed, only limited editing of device information is possible.) GUDID data is updated every business day, so the most up to date information can be searched or downloaded from this site.